Description
The Molecular Radiopharmacy team, linked to the Radiopharmacy sub-division, is actively engaged in the development and evaluation of radiolabeled vectors for oncological applications, based on the overexpression of target-biomolecules on cancer cells. Thus, following administration of GPCR-directed peptide radioligands in patients, visualization and/or eradication of cancer sites become feasible by means of diagnostic or therapeutic radiation specifically localized on tumor cells. Imaging is performed either by single photon emission computed tomography (SPECT), or by positron emission tomography (PET), whereas tumor cell apoptosis and death are induced by particle emitting radiation.
On one hand, our team has been developing GPCR-directed peptide radioligands carrying diagnostic or therapeutic radiometals to tumors and comprising: a) the targeting peptide, b) the chelator stably binding the radiometal of choice and c) a linker introduced between the two entities. On the other hand, it has been quite active in the preclinical evaluation of new peptide radioligands at the molecular and cellular level and in experimental animal models, which is the crucial link between molecule design and clinical application. Two major breakthroughs of the team are shown below (see: grants, infrastructures for more information):
- Marie Curie Award in 2013 for introducing the concept of improved tumor uptake of theranostic radiopeptides by their in situ stabilization in the circulation using key-protease inhibitors (https://www.eanm.org/congresses-events/awards-grants/winners/)

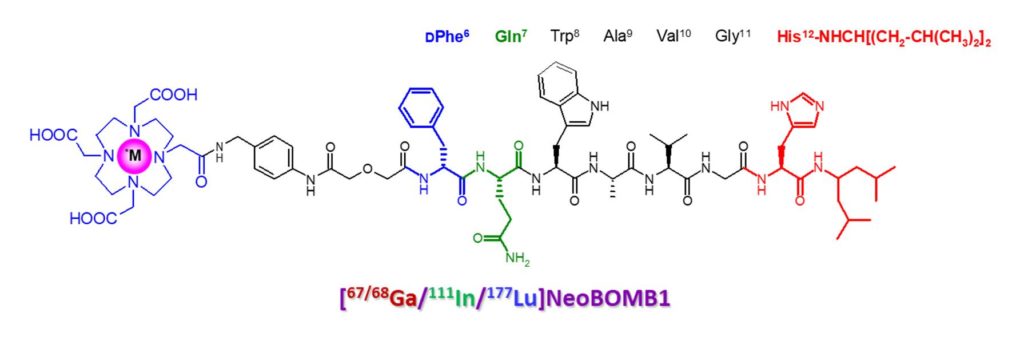
- Development of the theranostic [68Ga]Ga-/[177Lu]Lu-NeoB(omb) radiopeptide pair into a radiopharmaceutical used in the treatment of solid tumors in man, supported by pharmaceutical industry (https://www.adacap.com/pipeline/) and currently undergoing multicenter clinical trials (https://clinicaltrials.gov/ct2/show/NCT03872778?term=177lu&draw=2&rank=41).
People
Infrastructures
Latest Grants
Proud to Collaborate with

Netherlands

Netherlands

Germany

U.S.A.

Greece